hemoglobin
construction
Hemoglobin is a protein in the human body, which functions for the Oxygen transport of the blood owns. Proteins in the human body are always made up of multiple amino acids that are linked together. The body partially absorbs the amino acids into the body with food; the body can partially convert other molecules into amino acids through enzymatic conversions or produce them completely itself.
141 individual amino acids combine to form a subunit of hemoglobin, a globin. A hemoglobin molecule consists of four globins, with two identical subunits forming a molecule. The globins are folded in such a way that a kind of pocket is created in which a heme molecule, a so-called "iron complex", is bound. This iron complex, of which there are four in one hemoglobin molecule, binds one molecule of oxygen at a time, an O2.

Because of the iron in the structure, the hemoglobin takes on a red color, which gives all the blood its color. If the iron ion now binds an oxygen molecule, the color of the hemoglobin changes from a dark red to a lighter red. This change in color is also noticeable when comparing venous and arterial blood. The arterial blood, which carries more bound oxygen, has a significantly lighter color.
The four globin subunits have a special effect in binding the four oxygen molecules. With every oxygen molecule that is bound, interactions arise between the four subunits and the binding of another oxygen is facilitated. A hemoglobin loaded with four oxygen molecules is particularly stable. The delivery also works. Once one molecule of oxygen leaves the hemoglobin, the process is made easier for the other three as well.
Humans have different forms of hemoglobin in different life situations. As a child in the womb, he initially has embryonic and later fetal hemoglobin. The globin subunits differ in their chemical structure and ensure that hemoglobin in children has a significantly higher affinity for oxygen than adult hemoglobin. This enables the oxygen in the placenta to be transferred from the mother's to the child's blood.
The adult human can have two different types of hemoglobin, HbA1 or HbA2, but HbA1 predominates in 98% of all people.
If the blood sugar level remains too high for a long time, a sugar-coupled hemoglobin, the HbA1c, may be present. It is primarily used in diagnostics to analyze long-term blood sugar levels.
Methemoglobin is a non-functional form. It can no longer bind oxygen. It is present in small proportions in every person and is formed particularly strongly in the case of smoke poisoning or genetic defects. The higher its proportion, the greater the oxygen shortage for the human organism.
Function in the human body

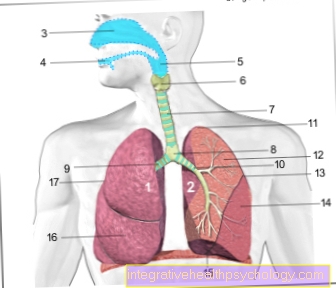
The function of the Hemoglobins is in the human body vital. The iron molecule in the center of the heme, which is carried by each globin subunit, binds an oxygen molecule. After the venous blood in the body is pumped from the right heart into the lungs, it accumulates there with the inhaled oxygen on. From then on it is called oxygen-rich. The oxygen diffuses over the boundaries of the alveoli through the vascular walls, into the red blood cells, the erythrocytes, and chemically binds to the iron ion. The blood takes on the typical light red, arterial color as a result of the bond and is then pumped through the body from the left heart via the large bloodstream. In the tissue that is supposed to supply the blood with oxygen, the blood flows particularly slowly through the capillaries so that the oxygen-poor tissue can withdraw the oxygen molecule from the oxygen-rich blood and the hemoglobin is converted back into its original form.
The effect of the "Cooperativity“Causes the four globin units to mutually simplify the loading and unloading of oxygen molecules. An oxygen bond that has already taken place makes the bond of the other three molecules much easier. This means that the oxygen content initially remains stable even with slight restrictions in oxygen enrichment. Even limitations in old age, when staying at high altitudes and slight pulmonary dysfunction do not initially have a strong influence on the oxygen saturation of the blood. Even if the Oxygen partial pressure has already dropped to half of the original value, the Oxygen saturation of blood still over 80%.
It is also very important that the hemoglobin property owns, depending on oxygen PH valueTo bind CO2 partial pressure, temperature and 2,3-BPG (2,3-bisphosphoglycerate) to different degrees. This makes it possible that as much as possible is bound in the lungs and that as much as possible can be released in the rest of the body tissue if it is needed. About the 2,3-BPG, which e.g. is increased during altitude training, the body can also reduce the binding strength of oxygen so that it can be released more easily.
In addition, the hemoglobin also has a certain function Degrees of CO2 to be transported and release it in the lungs. The carbon dioxide is also bound to the hemoglobin, but not to the O2 binding site.
The hemoglobin value is meaningful for many diseases. Especially the deficiency diseases, which are called Anemia are a common problem.
Hemoglobin too low
Since every red blood cell has the hemoglobin molecule, the hemoglobin value is a meaningful marker for the number of red blood cells in the bloodstream. A blood test can be carried out in medical laboratories to determine the Hb level and use this to estimate the number of red blood cells. If the value is below the normal range of the respective group of people, there is anemia, a so-called "anemia". A whole range of diseases and causes can be behind anemia. In most cases, however, the anemia can be treated well.
With the Hb value, additional values are found in the blood count, which often point to a cause of the anemia. These include the values MCH, MCHC, MCV and RDW. If the volume and hemoglobin content of an individual erythrocyte are reduced, the MCH, MCHC and MCV are below the normal range. In this case, an iron deficiency or a problem in iron metabolism are most likely. Iron deficiency anemia is a particularly common clinical picture, especially in women. Most of the causes are bleeding.
Read more on the subject below Erythrocyte parameters
Malnutrition and malnutrition are also a common cause of anemia. In rarer cases, genetic or other diseases are behind it. These include, for example, sickle cell anemia, thalassemia or malignant diseases such as myelodysplastic syndrome.
You can find out more about sickle cell anemia in our article Sickle Cell Anemia - How Dangerous Is It Really?
If the anemia is not acutely threatening, an attempt can first be made to remedy the cause. In the case of malnutrition, the focus is on nutritional advice. If the Hb levels are too low and pose risks in everyday life, the problem can be solved with one-off infusions. If there is an iron deficiency, it is sufficient to change the diet or to give the patient iron by infusion. In particularly acute situations, a blood transfusion may also be necessary. Blood transfusions are especially indicated after or during heavy bleeding or in the case of blood formation disorders. Regular blood transfusions are also necessary in patients with myelodysplastic syndrome.
The classic symptoms of anemia are paleness, tiredness, dizziness, and a feeling of weakness.
Hemoglobin too high
Since the hemoglobin value in the blood is indicative of the number of red blood cells, an increased value is often associated with an excessive number of erythrocytes. The blood is made up of solid and liquid parts, roughly 40:60.
The main part of the solid parts is found in the erythrocytes. If there are too many of them, one speaks of a "Polyglobules"Or"Erythrocytosis". Hb values that are too high are normal in people who have stayed at particularly high altitudes for a long time. Due to the reduced oxygen content at high altitudes, the body begins to produce additional oxygen carriers to compensate for the deficiency. Athletes take advantage of this effect by doing altitude training. Due to the increased number of erythrocytes, they are more efficient when exercising in normal oxygen conditions. This blood production can also be brought about with medication, but it is forbidden as "EPO doping" in sport.
Even long-term Smoker and patients with Lung disease often reactively have increased Hb values (hemoglobin values).
In some cases, the increased number of red blood cells can cause the so-called "Hematocrit value" increase. It describes the proportion of solid blood components (e.g. cells) in relation to the total blood. A greatly increased hematocrit can lead to Thrombosis, Heart attacks and Strokes to lead. Because of the many fixed components that is Blood thicker and flows more slowly through the vessels. A few such cases have been found in athletes who have committed enormous EPO doping. Strongly increased values bring this risk with them.
A pathological change that leads to increased hemoglobin levels in the blood is that Polycythemia vera. The disease is one of the myeloproliferative diseases, in which all solid blood components are increasingly produced, including the Leukocytes and Platelets. The disease is very rare and can be congenital or acquired.
If the Hb levels are slightly elevated, the patients usually do not notice any symptoms in everyday life. Anemia is a more common problem in everyday clinical practice than increased hemoglobin levels.
Hemoglobinopathy
Hemoglobinopathy is the umbrella term for diseases that cause changes in hemoglobin. These are genetically predisposed.
The best known are the Sickle cell anemia and the Thalassemias (divided into alpha- and beta-thalassemia). The diseases are either based on a mutation, i.e. a change, in the proteins (sickle cell anemia) or there is a reduced production of these (thalassemia).
The diseases have in common that they can be of different severity and that, depending on the severity, they lead to mild or severe anemia or even to infants who are unable to survive.
Hemoglobin in the urine
If there are hemoglobin molecules in the blood, this is a sign of a increased death of red blood cells in the bloodstream.This is known as Hemoglobinuria. The kidney filters the entire blood from the circulatory system several times within an hour. As a rule, however, it does not filter out proteins. If erythrocytes die within the blood vessels instead of in the spleen, where they are usually broken down, there are excessive hemoglobin molecules free in the blood. The kidneys then filter them out and excrete them in the urine. Similar to how hemoglobin gives blood its red color, The urine then also turns dark red.
Also malaria causes hemoglobinuria. However, certain types of Anemia or transfusion errors. If the cause is in the vascular system, hemoglobinuria is often present. It should not be confused with hematuria, in which whole blood is found in the urine. The reasons and causes for this are different.
HbA1C
The Hemoglobin A1C is a special form of hemoglobin in the human body. It is also known as glycohemoglobin and describes a normal hemoglobin molecule to which glucose is coupled is.
Everyone has a small amount of HbA1C in their blood, which can be determined with a blood test. It's in the blood especially a lot of sugar in the form of glucose, the conversion of hemoglobin into its “glycated” form takes place without enzymes. This process is not reversible. Since an erythrocyte survives on average eight weeks before it is broken down, the amount of HbA1C can be used to estimate the level of sugar in the blood over the last few weeks. The HbA1C value is therefore considered to be Blood sugar memory and is used in medicine.
Patients who have a known sugar disorder Diabetes mellitus should have their HbA1C level checked every three months. Of the Normal range is 4-6% Share in total hemoglobin. Diabetics should try to keep their value below 8% as well permanent damage by preventing the disease.
In patients with iron deficiency anemia, cirrhosis of the liver, renal insufficiency and other diseases that can affect the blood count, the values are falsified and inconclusive.
Standard values
The normal values for hemoglobin concentration differ from children to adults, but also between men and women. The reference ranges in adults Men are included 12.9 - 16.2 g / dl, at Women at 12-16 g / dl and at Newborn at 19 g / dl.
96% of all values for healthy people are in this range. However, when symptoms of anemia become noticeable, it varies from person to person. In everyday clinical practice, treatment is also made dependent on the clinical picture. Not every value outside the reference range indicates actual illness or anemia.
Hemoglobin synthesis
Mature erythrocytes no longer have a cell nucleus and can therefore no longer produce proteins. The hemoglobin, which is present in the red blood cells, thus becomes during the maturation of these in the stage of Erythroblasts (Pre-stage of red blood cells). As described above, hemoglobin consists of proteins and heme molecules, which are produced separately and then put together. It is important here that for the heme synthesis, i.e. production, both iron as well as Vitamin B6 are needed. This explains why a deficiency in these substances can lead to anemia.